
FDA Roundup: The Latest Activity
This week, FDA kicked-off a new resource about the Modernization of Cosmetics Regulation Act of 2002 (MoCRA) and a follow-up constituent advisory that FDA has

This week, FDA kicked-off a new resource about the Modernization of Cosmetics Regulation Act of 2002 (MoCRA) and a follow-up constituent advisory that FDA has
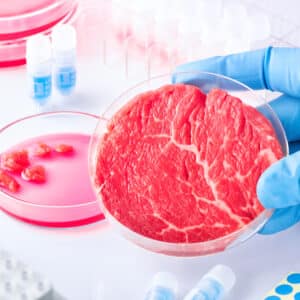
FDA completed its second pre-market consultation for human food made from cultured animal cells, and advised it had no further questions about the firm’s safety


FDA Issues Draft Labeling Recommendations for Plant-Based Milk Alternatives

Closing Gaps, Protecting Organic Producers and Improving Transparency and Traceability of Organic Products As the “organic” section of many grocery stores continues to grow, with

Protecting Consumers, Limiting Supply Chain Disruption and Avoiding Business and Reputation Damage Quickly and Effectively With product recalls consistently being in the news over undeclared

While the year is winding down, FDA remains active in enforcement and issuing new guidance. Here’s a look at what has been happening at the Agency.

FDA’s Proposed Rule for Amending Color Certification Fees: Written Comments due by January 3, 2023

A reminder to register or renew your food, dietary supplement, OTC drug, or medical device facility registration before December 31, 2022

Revisiting the Perks of FDA’s Voluntary Qualified Importer Program (VQIP)